Overview
Cardiol Therapeutics Inc. (NASDAQ: CRDL, TSX: CRDL) is a clinical-stage life sciences firm targeted on the analysis and scientific improvement of anti-inflammatory and anti-fibrotic therapies for the therapy of coronary heart illness. The Firm’s lead drug candidate, CardiolRx™ (cannabidiol) oral answer, is pharmaceutically manufactured and in scientific improvement to be used within the therapy of coronary heart illness. It’s acknowledged that cannabidiol inhibits activation of the inflammasome pathway, an intracellular course of recognized to play an vital position within the improvement and development of irritation and fibrosis related to myocarditis, pericarditis, and coronary heart failure.
Cardiol has obtained Investigational New Drug Utility authorization from america Meals and Drug Administration to conduct scientific research to judge the efficacy and security of CardiolRx™ in two illnesses affecting the guts: (i) a Section II multi-center open-label pilot research in recurrent pericarditis (irritation of the pericardium), which is related to signs together with debilitating chest ache, shortness of breath, and fatigue, and ends in bodily limitations, decreased high quality of life, emergency division visits, and hospitalizations; and (ii) a Section II multi-national, randomized, double-blind, placebo-controlled trial (the “ARCHER” trial) in acute myocarditis, an vital explanation for acute and fulminant coronary heart failure in younger adults and a number one explanation for sudden cardiac demise in individuals lower than 35 years of age.
Cardiol can be creating a novel subcutaneously administered drug formulation of cannabidiol meant to be used in coronary heart failure – a number one explanation for demise and hospitalization within the developed world, with related healthcare prices in america exceeding $30 billion yearly.
For extra details about Cardiol Therapeutics, please go to cardiolrx.com.
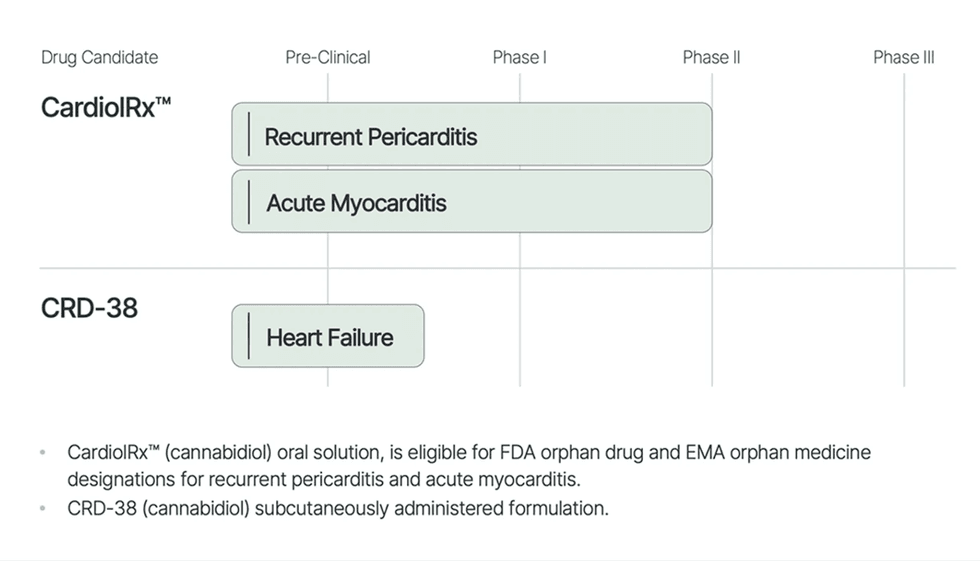
Firm Highlights
- Lead Asset in Scientific Growth: CardiolRx™, oral drug candidate, in Section II trials for recurrent pericarditis and acute myocarditis.
- Scientific Rationale: Compelling proof demonstrating the anti-inflammatory and anti-fibrotic properties of CardiolRx™ in myopericardial illnesses.
- Modern Analysis: Advancing the event of CRD-38, a novel proprietary subcutaneously administered pharmaceutical meant to be used in coronary heart failure.
- Broad Exclusivity Safety: Complete mental property portfolio. Eligible to pursue FDA orphan drug and EMA orphan drugs designations for CardiolRx™.
- Management: Skilled administration group, board of administrators, and scientific advisory board, with in depth experience in creating therapeutics for inflammatory coronary heart illness.
- Robust Monetary Place: Debt-free and well-capitalized to realize company milestones into 2026.
Key Initiatives
Orphan Drug Program for Recurrent Pericarditis
In January 2023, Cardiol introduced that the primary affected person has been enrolled within the company-sponsored Section II open-label pilot research investigating the tolerance, security, and efficacy of CardiolRx™ in sufferers with recurrent pericarditis. The research can even assess the development in goal measures of illness, and through an extension interval, assess the feasibility of weaning concomitant background remedy together with corticosteroids, whereas taking CardiolRx™.
Pericarditis refers to irritation of the membrane or sac that surrounds the guts (the pericardium) that’s most regularly triggered from a viral an infection. Recurrent pericarditis is the most typical complication following an preliminary acute episode of pericarditis, and sufferers could have a number of recurrences. Signs embrace debilitating chest ache, shortness of breath, and fatigue, leading to bodily limitations, decreased high quality of life, emergency division visits, and hospitalizations. Rare however life-threatening issues related to pericarditis embrace a big accumulation of pericardial fluid, scarring, and constriction of the guts which can restrict coronary heart operate. The illness is identified in 0.2% of all cardiovascular in-hospital admissions and is liable for 5% of emergency room admissions for chest ache in North America and Western Europe. Recurrent pericarditis is the re-appearance of signs after a symptom-free interval of a minimum of 4 – 6 weeks following an preliminary acute episode of pericarditis. These recurrences seem in 15% to 30% of acute circumstances and often inside 18 months. Moreover, as much as 50% of sufferers with a recurrent episode of pericarditis expertise extra recurrences. Commonplace first-line medical remedy consists of non-steroidal anti-inflammatory medication or aspirin with or with out colchicine. Corticosteroids equivalent to prednisone are second-line remedy in sufferers with continued recurrence and insufficient response to standard remedy. The one FDA-approved remedy for recurrent pericarditis, launched in 2021, is usually used as a third-line intervention in sufferers with a 3rd or fourth recurrence. The variety of circumstances of sufferers looking for and receiving therapy for recurrent pericarditis yearly within the U.S. is estimated at 38,000. Hospitalization on account of recurrent pericarditis is commonly related to a 5 – 8 day size of keep and value per keep is estimated to vary between $20,000 and $30,000 in america.

The Section II pilot research is anticipated to enroll 25 sufferers at scientific facilities in america focusing on pericarditis care. The protocol has been designed in collaboration with thought leaders in pericardial illness. The research chairman is Dr. Allan L. Klein, director of the Heart of Pericardial Ailments and professor of drugs, Coronary heart and Vascular Institute on the Cleveland Clinic. The first efficacy endpoint is the change, from baseline to eight weeks, in patient-reported pericarditis ache utilizing an 11-point numeric ranking scale (NRS). The NRS is a validated scientific instrument employed throughout a number of situations with acute and persistent ache, together with earlier research of recurrent pericarditis. Extra endpoints throughout extension interval embrace the NRS rating after 26 weeks of therapy, and adjustments in inflammatory marker C-reactive protein (CRP), a generally used scientific marker of irritation.
“We’re excited to be the primary scientific middle to manage this investigational drug in a affected person with recurrent pericarditis, a debilitating inflammatory coronary heart illness related to signs that adversely have an effect on high quality of life and bodily exercise,” commented Dr. Paul C. Cremer, heart specialist, Heart for the Prognosis and Remedy of Pericardial Ailments, Part of Cardiovascular Imaging, Division of Cardiovascular Drugs, Coronary heart, Vascular and Thoracic Institute, Cleveland Clinic, and research web site principal investigator. “Together with different collaborating analysis facilities all through the U.S., we sit up for full enrollment of members into this pilot research and to figuring out the potential of this remedy to deal with pericarditis and to scale back the danger of its recurrence.”
Within the US, an orphan drug designation is granted for prescription drugs being developed to deal with medical situations affecting fewer than 200,000 individuals. These situations are known as orphan illnesses. The task of orphan standing to a illness and to medication developed to deal with it’s a matter of public coverage in lots of nations and has yielded medical breakthroughs which may not in any other case have been achieved. Within the US and the European Union, orphan medication are eligible for accelerated advertising approvals and corporations creating orphan medication usually obtain different incentives, together with a chronic interval of market exclusivity that may lengthen over seven years, throughout which the drug developer has sole rights to market the drug.
Recurrent pericarditis is an orphan illness in america, thereby making CardiolRx™ eligible for orphan drug standing underneath the FDA’s Orphan Drug Designation program.
Impartial advisors and key investigators comprising six extremely distinguished thought leaders in cardiology from the Cleveland Clinic, the Mayo Clinic, the Monash Victoria Coronary heart Institute, and the College of Virginia, have been established to design, oversee, and information Cardiol’s Section II multi-center open-label pilot research in sufferers with recurrent pericarditis.

Orphan Drug Program for Acute Myocarditis
In August 2022, Cardiol enrolled its first affected person in ARCHER, the corporate’s multi-center, worldwide, double-blind, randomized, placebo-controlled trial designed to check the protection and tolerability of CardiolRx™, in addition to its affect on myocardial restoration, in sufferers presenting with acute myocarditis.
Myocarditis is an acute inflammatory situation of the guts muscle (myocardium) characterised by chest ache, impaired cardiac operate, atrial and ventricular arrhythmias, and conduction disturbances. Though the signs are sometimes gentle, myocarditis stays an vital explanation for acute and fulminant coronary heart failure and is a number one explanation for sudden cardiac demise in individuals underneath 35 years of age. Though viral an infection is the most typical explanation for myocarditis, the situation also can consequence from bacterial an infection, generally used medication and mRNA vaccines, in addition to therapies used to deal with a number of widespread cancers, together with chemo-therapeutic brokers and immune checkpoint inhibitors.
In a proportion of sufferers, the irritation within the coronary heart persists and causes decreased coronary heart operate with signs and indicators of coronary heart failure, and as such therapy relies on standard-of-care suggestions for coronary heart failure. This consists of diuretics, ACE inhibitors, angiotensin receptors blockers, beta blockers, and aldosterone inhibitors. For these with a fulminant presentation, intensive care is commonly required, with using inotropic drugs (to extend the power of the guts muscle contraction). Extreme circumstances regularly require ventricular help gadgets or extracorporeal oxygenation and will necessitate coronary heart transplantation.
There aren’t any FDA-approved therapies for acute myocarditis. Sufferers hospitalized with the situation expertise a median 7-day size of keep and a 4 – 6% danger of in-hospital mortality, with common hospital cost per keep estimated at $110,000 in america.

“The US orphan drug program was efficiently utilized to speed up the primary FDA approval of CBD for the therapy of uncommon types of pediatric epilepsy, and important shareholder worth was created within the course of,” said Cardiol President and CEO David Elsley. “Given the mortality and important morbidity danger related to acute myocarditis, we consider there’s a related alternative in pursuing an expedited improvement program of our CardiolRx™ pharmaceutical CBD formulation for this severe orphan illness which has no accepted customary of care.”
The first efficacy endpoints of the ARCHER trial encompass extracellular quantity (ECV) and world longitudinal pressure (GLS). The secondary efficacy endpoint is left ventricular ejection fraction. Since individuals with acute myocarditis have impaired coronary heart operate, present therapy relies on standard-of-care suggestions for coronary heart failure. This consists of diuretics, ACE inhibitors, angiotensin receptors blockers, beta blockers, and aldosterone inhibitors. For these with a extreme and sudden onset presentation, intensive care is commonly required, with using inotropic drugs (to extend the power of the guts muscle contraction) and sometimes, heart-lung bypass or ventricular help gadgets. There’s in any other case no particular therapy for acute myocarditis though some sufferers have responded to immuno-suppressive remedy (azathioprine) together with steroids, however the information usually are not conclusive sufficient for this to be the really useful remedy.
An impartial scientific steering committee, comprising 10 extremely distinguished thought leaders in cardiology from the Cleveland Clinic, the Mayo Clinic, the Houston Methodist DeBakey Coronary heart and Vascular Heart, the College of Ottawa Coronary heart Institute, McGill College Well being Centre, the College of Pittsburgh Medical Heart, College Drugs Berlin, Tel Aviv “Sourasky” Medical Heart, São Paulo College Medical College, and Pitié Salpêtrière Hospital (Sorbonne College), has been established to design, oversee, and information Cardiol’s Section II multi-national ARCHER trial in acute myocarditis.


Cardiol Therapeutics’ Coronary heart Failure Program
Coronary heart failure impacts greater than 64 million individuals globally and related healthcare prices exceed $30 billion yearly within the U.S. alone. Coronary heart failure is a persistent, progressive syndrome during which the guts muscle is unable to pump sufficient blood to satisfy the physique’s wants for blood and oxygen. Folks with coronary heart failure endure from shortness of breath, speedy coronary heart price, edema, decreased train capability, usually wrestle with easy day by day actions, and are regularly hospitalized. For a lot of, these signs considerably scale back their high quality of life. Identified causes of coronary heart failure embrace ischemic coronary heart illness and myocardial infarction (coronary heart assault), hypertension, valvular coronary heart illness, inflammatory illnesses of the guts equivalent to myocarditis and cardiomyopathies, anti-cancer therapies, and inherited metabolic illnesses.
Coronary heart failure stays a number one explanation for morbidity and mortality worldwide and persists as a rising well being and financial burden. In america alone, 6 million individuals over the age of 20 reside with coronary heart failure, and this quantity is projected to extend to >8 million by 2030. The overall annual price attributed to coronary heart failure is projected to extend to $69.8 billion by 2030. Latest reporting signifies that in america there are 3.3 million doctor visits with a major analysis of coronary heart failure yearly, and 1.5 million emergency division visits attributable to the syndrome. Whole deaths attributed to coronary heart failure yearly in america have been reported within the vary of 86,000 to >300,000, and hospitalizations vary from 800,000 to 1.3 million. The 5-year mortality price for these with coronary heart failure has been reported at 52.6% total.
Cardiol is creating CRD-38, a novel proprietary drug formulation designed to ship cannabidiol by subcutaneous administration. They’re endeavor IND-enabling actions to help scientific analysis of CRD-38 as a therapeutic technique in coronary heart failure care – a number one explanation for demise and hospitalization within the developed world, with related health-care prices in america exceeding $30 billion yearly. As well as, Cardiol has an energetic discovery program targeted on creating extra novel therapeutic approaches to deal with irritation and fibrosis related to the event and development of coronary heart illnesses.
Revealed third-party analysis has proven that cannabidiol reduces inflammatory activation of the endothelial lining of blood vessels and aids endothelial vasorelaxation, leading to improved blood circulation. Cannabidiol has additionally been proven to attenuate quite a lot of measures of irritation in fashions of diabetes, a standard comorbidity in coronary heart failure sufferers, and to scale back myocardial fibrosis in a mannequin of inflammatory coronary heart illness.
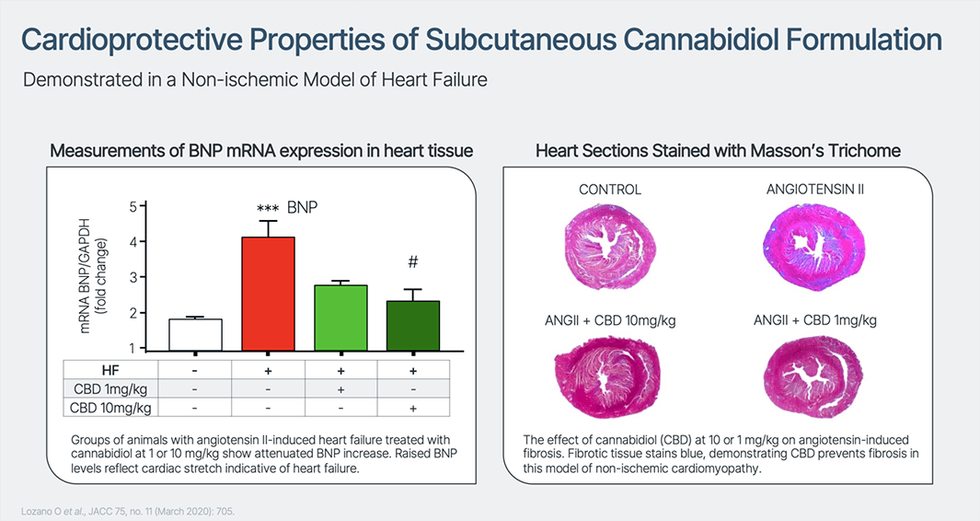
Cannabidiol is lipid soluble, just about insoluble in water, extremely delicate to deactivation within the liver by way of first-pass metabolism when taken orally and is quickly cleared from the physique. This ends in a low total bioavailability when taken orally. Cardiol’s subcutaneously administered drug formulation is designed to attenuate first-pass metabolism, optimize and keep blood ranges of the drug, and goal irritation and elevated fibrosis within the coronary heart. Cardiol believes that overcoming the low bioavailability points related to cannabidiol will considerably broaden the therapeutic potential of this molecule.
Cardiol Therapeutics’ Key International Analysis and Scientific Collaborators
Cardiol is working along with world-class researchers and clinicians at worldwide facilities of excellence to leveraging their experience in drug improvement, experimental execution, irritation and fibrosis, the therapy of cardiovascular illnesses, and scientific trial protocol design. The collaborations present optimum recommendation and information platform in pursuit of Cardiol’s function: heal the guts with revolutionary science

Administration Crew
David Elsley – President, Chief Government Officer, and Director
David Elsley is the founder and CEO of Vasogen Inc. and has an MBA diploma. Elsley has over 25 years of expertise creating, financing, and managing all features of company improvement in biotechnology and high-growth organizations. Elsley based Vasogen Inc., a biotechnology firm targeted on the analysis and industrial improvement of novel therapeutics for the therapy of coronary heart failure and different inflammatory situations. Elsley assembled a group of administration, administrators and scientific advisors comprising trade professionals and thought leaders from North America and Europe.
Dr. Andrew Hamer – Chief Medical Officer and Head of Analysis and Growth
Dr. Andrew Hamer has an MBChB diploma. He’s the previous govt director at Amgen, liable for main world improvement of Repatha®. Hamer is the previous chief heart specialist at Nelson Hospital, New Zealand. He has over 19 years of expertise working towards cardiology and inner drugs.
Chris Waddick – Chief Monetary Officer and Director
Chris Waddick has an MBA diploma, is a chartered skilled accountant, and is an authorized administration accountant. He has over 30 years of expertise in monetary and govt roles within the biotechnology and power industries. Waddick is the previous chief monetary officer and chief working officer of Vasogen Inc.
Bernard Lim – Chief Working Officer
Bernard Lim has over 30 years of expertise within the life sciences trade, spanning biotechnology, diagnostics, medical gadgets, and high-technology corporations. He’s the founder and CEO of a extremely profitable drug supply firm that he led from analysis and improvement via to commercialization, and facilitated its eventual acquisition by Eli Lily. Lim is a chartered engineer per UK requirements and is a member of the establishment of engineering and expertise.
Andrea B. Parker – Senior Director of Scientific Operations
Dr. Andrea Parker is the previous chief scientific officer at Peter Munk Cardiac Heart, College Well being Community. Parker is a scientific epidemiologist with greater than 30 years’ expertise in scientific trials design, administration, and execution in trade and tutorial settings.
John A. Geddes – Vice-president, Enterprise Growth
John Geddes has over 25 years of expertise within the healthcare trade, comprising roles inside pharmaceutical, biotechnology, scientific diagnostics, and life science analysis expertise corporations. Geddes has an MBA diploma and is the previous company senior director, enterprise improvement at Luminex Company, a DiaSorin Firm.
Anne Tomalin – Director of Regulatory and High quality
Anne Tomalin is the founding father of CanReg and TPIreg, regulatory corporations beforehand bought to Optum Perception and Innomar Methods, respectively. Tomalin is an knowledgeable in regulatory affairs in Canada, america, and Europe.
Board of Administrators
Guillermo Torre-Amione – Chairman
Guillermo Torre is the president of TecSalud tutorial medical middle and faculty of the Instituto Tecnológico y de Estudios Superiores de Monterrey (ITESM), Mexico. He’s the previous director of Cardiac Transplantation on the Houston Methodist DeBakey Coronary heart & Vascular Heart.
Jennifer M. Chao – Director
Jennifer M. Chao has over 25 years of expertise within the biotech and life sciences industries targeted totally on finance and company technique. Chao is Managing Companion of CoreStrategies Administration, an organization she based in 2008 to supply transformational company and monetary methods to biotech/life science corporations for maximizing core valuation. She presently serves on the board of administrators of Endo Prescribed drugs and is a member of the audit committee and compliance committee. Previous to becoming a member of Endo, Chao served as chairman of the board of BioSpecifics Applied sciences from October 2019, till its acquisition by Endo for roughly US$660 million in December 2020. She additionally served as chair of BioSpecifics’ compensation committee and as a member of the audit committee, technique committee, mental property committee, and nominating and company governance committee from 2015 to 2020.
Peter Pekos – Director
President and CEO at Dalton Pharma, Peter Pekos has broad expertise in analysis, improvement, and commercialization of prescription drugs, merchandise, and companies.
Colin Stott – Director
Colin Stott has over 30 years of expertise in pre-clinical and scientific improvement, with particular experience within the improvement of cannabinoid-based medicines. Stott is the chief working officer of Alterola Biotech Inc. and the previous scientific affairs director, worldwide, and analysis and improvement operations director for GW Prescribed drugs, a world chief within the improvement of cannabinoid therapeutics.
Teri Loxam – Director
Over 25 years of expertise within the pharmaceutical, life sciences, and TMT industries with various roles spanning technique, investor relations, finance, and communications. Loxam joined Gameto, a biotechnology firm utilizing cell engineering to develop therapeutics for illnesses of the feminine reproductive system, in April 2023 as chief monetary officer. On this position, Loxam oversees monetary operate, in addition to performs a key position in total firm technique. Previous to becoming a member of Gameto, Loxam was chief working officer and chief monetary officer at Kira Prescribed drugs. Previous to becoming a member of Kira, Loxam served as chief monetary officer at SQZ Biotech the place she led the corporate’s monetary operations, investor relations and communications/public relations capabilities. Previous to becoming a member of SQZ, Loxam held varied positions at Merck, IMAX Company, and Bristol-Myers Squibb throughout communications, technique, treasury, and investor relations.
Michael Willner – Director
Michael Willner has practiced as each an lawyer and an authorized public accountant. He graduated from Emory College Regulation College as a member of the Emory Regulation Overview. Subsequently, he practiced actual property and company legislation with New York Metropolis-based Milbank, Tweed, Hadley & McCloy, one of many nation’s most distinguished worldwide legislation corporations. Previous to his authorized profession, Willner was employed by the previous Arthur Andersen & Firm, a nationwide accounting agency, the place he practiced within the tax division.
Scientific Advisory Board
Dr. Paul Ridker
Dr. Paul Ridker is director of the Heart for Cardiovascular Illness Prevention, a translational analysis unit at Brigham and Ladies’s Hospital in Boston (BWH). A cardiovascular drugs specialist, he’s additionally the Eugene Braunwald Professor of Drugs at Harvard College of Drugs (HSM). Ridker obtained his medical diploma from HSM, after which accomplished an inner drugs residency and a cardiology fellowship at BWH. He’s board licensed in inner drugs. Ridker’s scientific pursuits embrace coronary artery illness and the underlying causes and prevention of atherosclerotic illness. He’s the creator of over 900 peer-reviewed publications and evaluations, 64 guide chapters, and 6 textbooks associated to cardiovascular drugs.
Dr. Bruce McManus
Dr. Bruce McManus is professor emeritus of the Division of Pathology and Laboratory Drugs on the College of British Columbia. He has served as CEO of the Heart of Excellence for Prevention of Organ Failure (PROOF Heart), director of the UBC Heart for Coronary heart and Lung Innovation, and scientific director, Institute of Circulatory and Respiratory Well being, CIHR. McManus obtained BA and MD levels from the College of Saskatchewan, an MSc from Pennsylvania State College, and a PhD from the College of Toledo. McManus pursued post-doctoral fellowships on the College of California, Santa Barbara in environmental physiology and on the Nationwide Coronary heart, Lung, and Blood Institute in Bethesda. McManus served as MD in cardiovascular and pulmonary pathology, and accomplished residency coaching on the Peter Bent Brigham Hospital, Harvard College, in Inner Drugs and Pathology.
Dr. Joseph Hill
Dr. Joseph Hill is a professor of inner drugs and molecular biology, chief of cardiology at UT Southwestern Medical Heart, in Dallas, and is the director of the Harry S. Moss Coronary heart Heart. Hill holds each the James T. Willerson, MD, distinguished chair in cardiovascular illnesses, and the Frank M. Ryburn Jr. Chair in Coronary heart Analysis. He graduated from Duke College with an MD and a PhD in 1987. Hill’s PhD dissertation analysis was within the subject of cardiac ion channel biophysics. He then labored for 5 years as a postdoctoral fellow on the Institut Pasteur in Paris, learning central and peripheral nicotinic receptors. He subsequent accomplished an inner drugs internship and residency, in addition to a scientific cardiology fellowship, on the Brigham and Ladies’s Hospital, Harvard Medical College.




